Colorectal Cancer Screening
How do they screen for colorectal cancer?
There are several different tests that doctors use to screen for colorectal cancer. National guidelines recommend who should consider colorectal cancer screening, which screening options are most appropriate and at what age screening should start. These guidelines vary based on gene mutation, family history and other factors. See below for more information and talk with your doctor to decide which screening and schedule is right for you.
Symptoms of colorectal cancer
Everyone should be aware of the symptoms of colorectal cancer, especially people at high risk. Not all colorectal cancers cause symptoms, this is why screening is so important. You should report any of the following to your doctor:
- changes in your bowel habits
- blood in or on your stool
- diarrhea, constipation, or feeling that the bowel does not empty all the way
- belly pain or cramps that don't go away
- unexplained weight loss
The symptoms listed above can be caused by many things, including colorectal cancer. Your doctor can run additional tests to try to determine the cause of your symptoms.
Types of colorectal cancer screening
Most colorectal cancers start as an abnormal growth known as a polyp. Although most will never turn into a colorectal cancer, some do have the potential to grow into cancers. The goal of colorectal cancer screening is to find and growths and remove them before they have the chance to turn into cancer. If cancer has already formed, early detection can still help improve a person’s chance of detecting cancer at an earlier, more treatable .
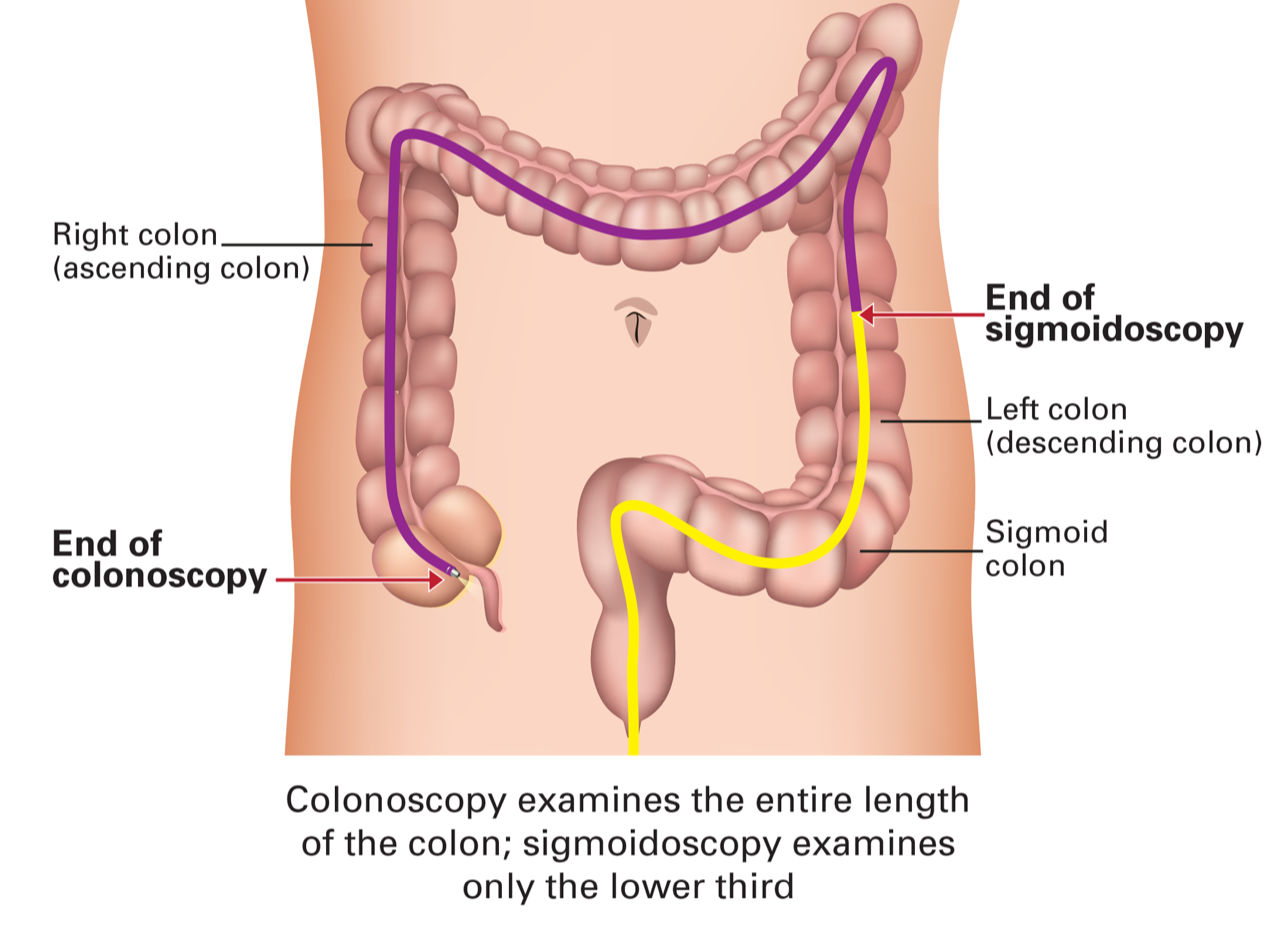
 Colonoscopy
Colonoscopy
A colonoscopy involves a doctor using a long, flexible tube with a small video camera attached to look at the entire length of the colon and rectum. This exam is done under sedation while the doctor inserts the tube through the anus into the rectum and into the colon. The scope has openings for instruments the doctor can use to remove any , which can be examined under a microscope to look for cancer.
Preparation for a colonsccopy begins 1-2 days before the exam to empty the colon of any stool. This allows the doctor to get a good look at the lining of the entire colon. The doctor prescribes a combination of a liquid diet, laxatives (usually taken as pills or large amount of liquid) and in some cases, enemas.
Colonoscopy is the most effective screening tool for colorectal cancer. It can actually prevent many cases of colorectal cancer by finding and removing before they can become cancer.
 Flexible sigmoidoscopy
Flexible sigmoidoscopy
Flexible sigmoidoscopy is similar to colonoscopy, using a flexible tube with a video camera to examine the colon and rectum. The scope has openings for instruments the doctor can use to remove any , which can be examined under a microscope to look for cancer. However, the scope used for sigmoidoscopy is shorter than the scope used for colonoscopy. It can only examine the rectum and a small part of the colon. Because the scope is shorter than the one used for colonoscopy, it may miss more aggressive cancers that can develop in the right side of the colon (first part of the colon).
As with colonoscopy, sigmoidoscopy requires preparation to empty the colon beginning 1 day before the exam. Most people don't need sedation for sigmoidoscopy.
Colonography
This test uses a type of xray scan known as a to take detailed, 3-dimensional pictures of the colon and rectum. This test is not as invasive as a colonoscopy, however, it does involve filling the colon and rectum with air using a small, flexible tube. The goal of the scan is to find any . If or abnormalities are found during colonography, a followup colonoscopy is needed to remove the .
This test does not require any sedation. However, the same type of bowel preparation needed for a colonoscopy or sigmoidoscopy is needed 1-2 days before the exam.
Stool tests
Stool tests look for possible signs of colorectal cancer—such as blood or abnormal —in stool samples. There are several different tests available. These tests require a doctor's prescription. The test kit is sent by mail and comes with simple instructions on how to collect the sample and mail it back to the lab. If abnormalities are found during stool tests, a followup colonoscopy is needed to find and remove any .
Colorectal cancer screening guidelines
Experts have different recommendations for screening based on a person's risk for colorectal cancer. There are separate guidelines for screening people with an increased risk versus people who are high risk due to an or family history of colorectal cancer.
Colonoscopy screening for people with inherited mutations
Screening guidelines for high risk people involve colonoscopy. The recommended age for beginning screening depends on the gene mutation and, in some cases, personal or family history of cancer or . The NCCN guidelines include screening recommendations for people with inherited mutations in the genes listed below.
|
Gene |
Beginning Age |
Frequency |
|
20 - 25 (or 2-5 years before the earliest age of onset if younger than 25). |
Every 1-2 years. (See , and risk-management pages for more extensive information). |
|
|
30 - 35 (or 2-5 years before the earliest age of onset if younger than 30). |
Every 1-3 years. (See and risk-management pages for more extensive information). |
|
|
10 - 15 |
Annually. (See (FAP/AFAP) risk-management page for more extensive information). |
|
|
AXIN2, GREM1, MSH3 (two mutations/MSH3-associated polyposis), NTHL1 (two mutations) |
25 - 30 |
Every 2-3 years if no found. If are found, every 1–2 years. Consider risk-reducing surgery if too many to manage with colonoscopy. |
|
12 - 15, or earlier if showing symptoms. |
Every 2-3 years or more frequently based on findings. |
|
|
25 - 30 |
Every 1-2 years. (See MUTYH (MAP) risk-management page for more extensive information). |
|
|
25 - 30 (or 2-5 years before the earliest age of onset if younger than 25). |
Every 2-3 years if no found. If are found, every 1–2 years. Consider risk-reducing surgery if too many to manage with colonoscopy. |
|
|
35 |
Every 5 years. (See risk-management page for more extensive information). |
|
|
18 |
Every 2–3 years or more frequently depending on number, size and type of found.. (See risk-management page for more extensive information). |
|
|
25 |
Every 2–5 years. (See risk-management page for more extensive information). |
|
|
Source: NCCN Guidelines: Genetic/Familial High-Risk Assessment: Colorectal, Endometrial, Gastric v. 1, 2025. |
||
Age to start screening and type of screening for people at high risk due to family history
The National Comprehensive Cancer Network (NCCN) has specific screening guidelines for people at high risk based on their family history of colorectal cancer.
|
Family History |
Age to Start |
Guideline |
|
with colorectal cancer at any age |
40 or 10 years earlier than the earliest colorectal cancer in the family |
Colonoscopy every 5 years or sooner depending on findings |
|
Second-degree and third-degree relative with colorectal cancer at any age |
45 |
Colonoscopy every 10 years or sooner depending on findings |
|
with certain types of advanced (based on pathology of polyp) |
40 or age of onset of advanced polyp in relative |
Colonoscopy every 5-10 years or sooner depending on findings |
|
Source: NCCN Guidelines: Colorectal Cancer Screening, Vs. 2, 2025. |
||
Screening for people with average risk
The U.S. government and many health organizations have recommendations for colorectal cancer screening and other preventative measures. Recommendations from the American Cancer Society (ACS), the National Comprehensive Cancer Network (NCCN) and the U.S. Preventive Services Task Force () are shown below.
Age to start screening for average-risk people
|
Screening Recommendation |
ACS |
NCCN |
|
|
Begin routine colorectal cancer screening for people at average risk. |
45 |
45 |
45 |
|
Discontinue routine screening. |
Screening from ages 76-85 should be based on personal preferences, health status and prior screening history. |
Screening from ages 76-85 should be based on personal preferences, health status and prior screening history. |
75 |
|
Source: NCCN Guidelines: Colorectal Cancer Screening, Vs. 2, 2025; American Cancer Society, Colorectal Cancer Screening (retrieved from website - 10/1/2024); US Preventive Services Task Force Colorectal Cancer: Screening, 2021 (retrieved from website 10/1/2024). |
|||
|
Type of Test |
Recommended Frequency |
|
Fecal protein test (FIT) |
Yearly |
|
Fecal blood test (gFOBT) |
Yearly |
|
Fecal test (FIT-DNA) |
Every 1-3 years |
|
Colonoscopy |
Every 10 years |
|
CT colonography |
Every 5 years |
|
Flexible sigmoidoscopy |
Every 5 years |
|
Flexible Sigmoidoscopy with FIT |
Flexible Sigmoidoscopy every 10 years plus FIT every year |
Paying for colorectal cancer screening
Under the Affordable Care Act most health insurance companies must pay for colorectal cancer screening as recommended by the United States Preventive Services Task Force with no out-of-pocket costs.
- Most health plans must cover 100% of one colorectal cancer screening at its recommended frequency with no out-of-pocket costs to patients who are age 45 and older—no matter their risk.
- Medicare beneficiaries—regardless of their age—are allowed one colonoscopy covered at 100% every 6 years for those at average risk and one colonoscopy per 24 months for those at high risk.
- Medicaid coverage of colorectal cancer screening varies by state. Individuals who qualify based on their state’s decision to expand Medicaid under the ACA are entitled to the same screening and preventive services as those who are covered by private insurance.
For individuals with increased risk, certain states require insurance coverage for colonoscopy beyond that required under the ACA. Check with your state insurance commission to determine if you live in one of these states.