Breast Cancer Stages & Subtypes
Measuring breast cancer stages and subtypes
After a biopsy or surgery confirms breast cancer, a pathologist will look at the cell type and perform tests for additional clues on how to best treat the cancer. Additional tests will be ordered to look for cancer spread through the local tissue or to distant organs.
Cell types
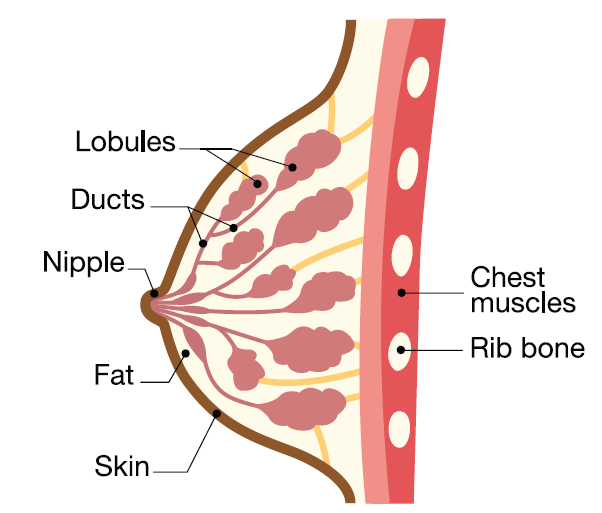
Breasts are made up of two main types of cells; the breast lobules, which make milk, and the breast ducts which carry the milk to the nipple.
- Ductal carcinoma is cancer that starts in the ducts. This is the most common type of breast cancer.
- Lobular carcinoma starts in the breast lobules. This is less common than ductal carcinoma.
In general, ductal carcinoma is more common than lobular carcinoma. However, people with inherited mutations in the gene are more likely to be diagnosed with lobular carcinoma.
Stages
The of a cancer refers to whether the cancer has spread beyond the breast, and if so, where in the body the cancer has spread. Measuring the of breast cancer helps doctors decide how to treat it. Breast cancer most often starts as a lump within the breast that can grow and spread:
- within the breast
- to nearby tissue (such as , skin or the chest wall)
- through the blood stream or lymph system to distant organs ()
During surgery, doctors will usually check one or more in the armpit to see if the cancer has spread. Doctors may also order tests such as chest XRAYS, , CT scans, bone scans or PET scans to learn if the cancer has spread beyond the breast and local lymp nodes.
In breast cancer, there are five major stages.
|
|
Description
|
|
0 ()
|
A 0 breast cancer is known as a ductal carcinoma in situ (), a non-invasive form of the disease. The tumor cells are contained within the ducts of the breasts, and have not invaded beyond the duct. Cancers caught and treated at 0 are very unlikely to recur or spread.
|
|
1
|
A small tumor (less than 2 cm) that is contained within the breast and has not spread to the .
|
|
2
|
These cancers may be either: a) a larger tumor that involves the breast only, or b) a tumor that has spread to some in the armpit.
|
|
3
|
These breast cancers may be either a) a tumor in the breast that invades the skin or chest muscle, or b) a tumor that involve several surrounding the breast (in the armpit, near the collarbone, near the breast bone). Some 3 breast cancers are considered “locally advanced.”
|
|
4 ()
|
These breast cancers have spread beyond the breast and the regional to another place in the body, such as the bone, lung, and/or liver. IV breast cancers are also called “advanced” or “metastatic.”
|
|
Stages of breast cancer
|
|
Most breast cancers diagnosed at stages 0-3 are considered "," because they are curable. breast cancer is often treatable at diagnosis. Over time, breast cancer is likely to progress or recur; sometimes after several different types of treatment. There are many different treatments available for breast cancer, and clinical trials testing new treatments are often available.
Subtypes and biomarkers
After a biopsy or surgery confirms breast cancer, pathologists look closely at the cells for additional clues on how to best treat the cancer. All breast cancers are tested for common changes, called tumor biomarkers: receptor (ER), progesterone receptor (PR), and . You can read more about testing and these subtypes.